
So we can substitute this value in for ?y?, and then simplify the decay formula. But regardless of the substance, when we’re looking at half life, we know thatīecause ?y? is the amount of substance that remains as the substance decays, and because ?C? is the amount of substance we started with originally, when the substance has decayed to half of its original amount, ?y? will be equivalent to ?C/2?. Because every substance decays at a different rate, each substance will have a different half life. Since substances decay at different rates, ?k? will vary depending on the substance.Įvery decaying substance has its own half life, because half life is the amount of time required for exactly half of our original substance to decay, leaving exactly half of what we started with. Where ?C? is the amount of a substance that we’re starting with, ?k? is the decay constant, and ?y? is the amount of the substance we have remaining after time ?t?.

So, for now, we’ll just state that the basic equation for exponential decay is
Calculate half life how to#
*The amount is listed in Ci ( Curies), but the mass, intensity of radiation, number of counts or other ways of characterizing the radioisotpe will decrease by the same ratio, so the calculation can be used for any of those quantities.We won’t work through how to prove these formulas, because in addition to derivatives, we also use integrals to build them, and we won’t learn about integrals until later in calculus. If you enter a fraction or amount remaining, it will calculate the time required (or age) required to achieve that fraction. Then if you enter a time, then it will calculate the fraction remaining at that time. It is presumed that you will enter a half-life for a particular radioisotope. time (t), i.e., the length of half-life increases with the decrease of concentration of the substrate. This calculation is designed for exploration. Half-life calculation on second order reactions results in concentration A vs. If the beginning amount was Ci* then the amount remaining is Ci If the radioactive half-life of an isotope isĪnd amount A 0 is present at time t=0 and amount A remains at time t, then
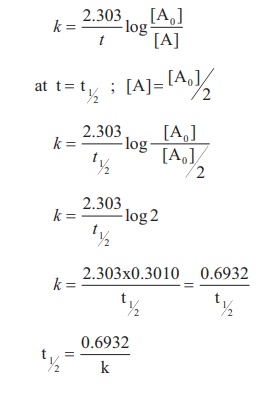
The calculation below is stated in terms of the amount of the substance remaining, but can be applied to intensity of radiation or any other property proportional to it. We can still calculate the amount of material we have left. The number of atoms, the mass of the substance, and the level of activity all follow the same exponential decay form. An interesting and useful aspect of radioactive decay is half-life, which is the amount of. Alternatively, this information can be expressed in terms of the decay constant λ. The radioactive half-life for a given radioisotope is a measure of the tendency of the nucleus to "decay" or "disintegrate" and as such is based purely upon that probability. Radioactive Half-Life Radioactive Decay Calculation The half-life is determined from the fundamental definition of activity as the product of the radionuclide decay constant,, and the number of radioactive.
